Ion exchange, as a traditional
process, has been around since the time of the Romans. The best known
application of ion exchange is the softening of water to remove calcium and
other scaling agents. Ion exchangers are solid adsorbent materials that have
charge sites containing exchangeable cations or anions. Ion exchange is a
stoichiometric and diffusion-rate-controlled process whereby cations and anions
can be interchanged from the solution phase to the solid exchanger phase.
Conventional batch ion exchange utilizes large vessels filled with ion exchange
resins. These systems are usually wasteful in consumption of water and
chemicals and so historically have been restricted to applications where feeds
are diluted or where product values are extremely high.
Adsorption is similar to ion exchange in that
species in solution transfer to the solid phase and in a cyclic process a
change in solution properties can be used to desorb the species in a
regeneration step.
State of the art . Over the last two decades,
significant advances have occurred in the field of solid adsorbents and
exchangers and even more so in the field of contacting with the advent of new
contactor technology.
|
|
Continuous contactors - The equipment advances
in the last 20 years have led to 2nd and 3rd generation continuous systems for
adsorption and ion exchange. These advances extend the economic range for
separation & purification applications through reduction in operating and
investment costs. Much less water and chemical consumption reduces the running
cost while faster cycles are driving space requirements and equipment costs to
new lows. The IXSEP system is an example of truly advanced continuous
contacting.
Resin beads - Polymer beads can be functionalized with many
new and selective chemical groups making them more specific separators. Polymer
structure advances make them kinetically faster and mechanically more robust.
|
|
|
|
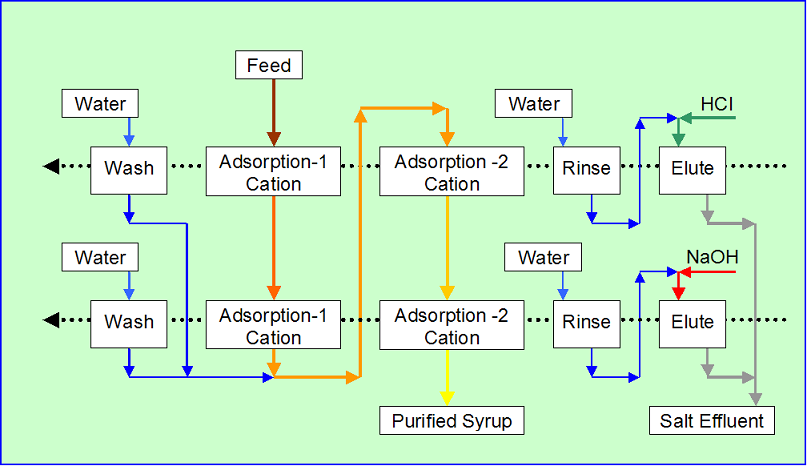
Example
|
|

|
|
|
 |
Ionex Separations, a division of Total
Project Solution, Inc.
About Us
|